Background Reading
Background: What is a polymer?
Exerpted from Brittanica Kids https://kids.britannica.com/students/article/polymer/276496
Exerpted from Kids Encyclopedia Facts. https://kids.kiddle.co/Polymer
The term polymer is a composite of the Greek words “poly” and “meros”, meaning “many parts.” Polymers are large molecules made of small, repeating molecular building blocks called monomers. The process by which monomers link together to form a molecule of a relatively high molecular mass is known as polymerization. Many polymer molecules are like chains where the monomer units are the links. Polymer molecules can be straight-chains or they can have cross-linking between chains.
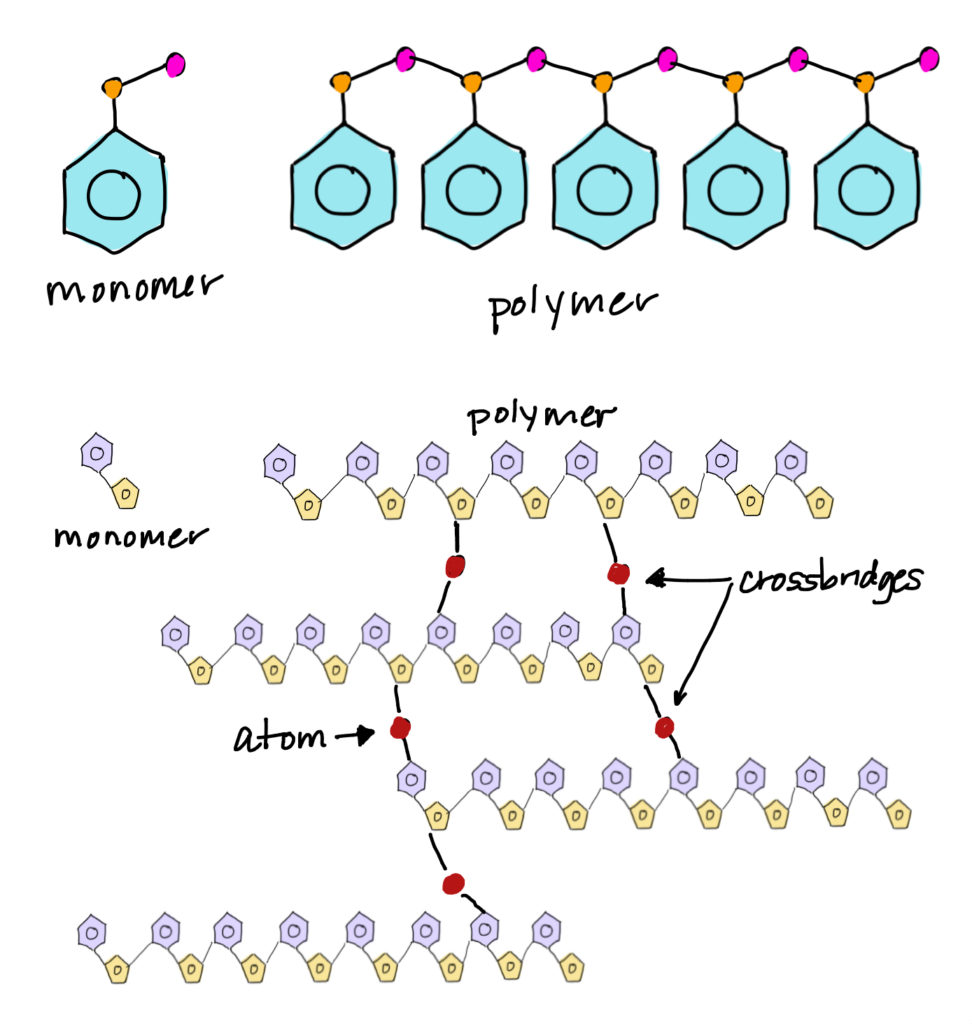
Polymers make up many of the materials in living organisms. Proteins are polymers of amino acids, cellulose is a polymer of sugar molecules, and nucleic acids such as deoxyribonucleic acid (DNA) are polymers of nucleotides. Many synthetic materials, including nylon, paper, plastics, and rubbers, are also polymers.
A variety of simple molecules join together to become useful polymers. The nonstick cookware coating known as Teflon, for example, is made of a monomer composed of two atoms each of fluorine and carbon. Both Plexiglas and Lucite are made of methyl methacrylate, an organic monomer composed of carbon, hydrogen, and oxygen. Silicon polymers used for sealants and other applications are made from inorganic monomers that contain silicon atoms.
Hydrogels are man-made substances that absorb water and hold it in the form of a gel. They’re used in disposable diapers, sanitary pads, breast pads, wound dressings, breast implants, and contact lenses. Hydrogels are also used to thicken products such as bubble bath and lotions and give them a silky, moisturizing feel.
Today, we will be looking at the polymers that make cross-linking chains and are able to hold water. They are called super-absorbant polymers (SAP) and hydrogels.
Do some additional research by exploring “Polymer Science Learning Center” https://www.pslc.ws/macrog/kidsmac/basics.htm

